While the Ramblings have recently being posted later than the first day shift, I’m still trying to keep posting a new article during every Red Shift’s set of four – but in some weeks the jostle for time between family life and other projects mean I could actually do with a 30 hour day. For this Rambling I’m killing two birds with one stone, the research undertaken for Susie and I to write an Education Toolkit for one of our Metro hospital’s ED departments will be on the same subject as this weeks Ramblings – prolonged QT. This is a subject we should especially be aware of as ECG changes including QT interval prolongation have been observed in patients receiving ondansetron. In addition, Torsade de Pointes, an abnormal, potentially fatal, heart rhythm, has been reported in some patients receiving ondansetron.
The QT interval is the time from the start of the Q wave to the end of the T wave. It represents the time taken for ventricular depolarisation and repolarisation, effectively the period of ventricular systole from ventricular isovolumetric contraction to isovolumetric relaxation. The QT interval is inversely proportional to heart rate:
- The QT interval shortens at faster heart rates;
- The QT interval lengthens at slower heart rates;
- An abnormally prolonged QT is associated with an increased risk of ventricular arrhythmias, especially Torsades de Pointes; and
- Congenital short QT syndrome has been found to be associated with an increased risk of paroxysmal atrial and ventricular fibrillation and sudden cardiac death.
The evaluation of every electrocardiogram should also include an effort to interpret the QT interval to assess the risk of malignant arrhythmias and sudden death associated with an aberrant QT interval. However, the correct determination of the QT interval, and its value, can appear to be a daunting task. Although computerised analysis and interpretation of the QT interval are available, these may over- or underestimate the QT interval.
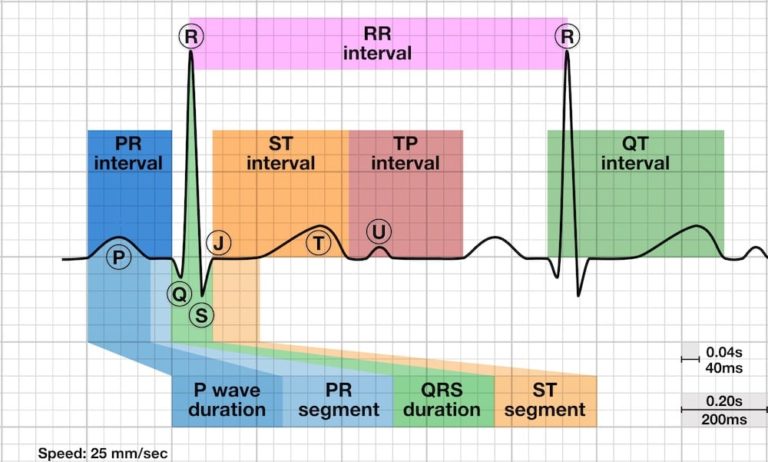
The QT interval is different in different leads (as all ECG parameters are). This is caused by the varying projections on different lead vector axes. Historically, measurement of P-QRS-T intervals are usually performed in lead II. There are several reasons for this custom: first, in the earliest years of the ECG there were no precordial leads and lead choice was thus confined to the limb leads. Furthermore, the vector axis of the P, QRS and TU-wave is predominantly directed infero-lateral and thus in the direction of lead II, resulting in easily recognisable P, QRS, T and U-waves in lead II, which promotes their measurement. The QT interval should be measured in either lead II or V5-6 if the T-wave is not easily measurable in lead II.
- Several successive beats should be measured, with the maximum interval taken;
- Large U waves (> 1mm) that are fused to the T wave should be included in the measurement;
- Smaller U waves and those that are separate from the T wave should be excluded; and
- The maximum slope intercept method is used to define the end of the T wave.
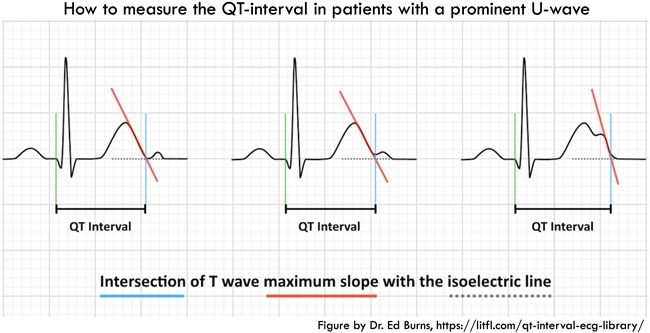
The QT interval is defined from the beginning of the QRS complex to the end of the T wave. The maximum slope intercept method defines the end of the T wave as the intercept between the isoelectric line with the tangent drawn through the maximum down slope of the T wave (pictured). A guide to measuring the QT interval can be found here:
https://www.cidg.org.nz/wp-content/uploads/2017/08/How-to-measure-a-QT-interval.pdf
As the QT interval varied with heart rate, the corrected QT interval (QTc) estimates the QT interval at a standard heart rate of 60 bpm. This allows comparison of QT values over time at different heart rates and improves detection of patients at increased risk of arrhythmias.
- QTc is prolonged if > 440ms in men or > 460ms in women;
- QTc > 500 is associated with increased risk of torsades de pointes, is clinically significant and likely to confer an increased risk of arrhythmia. Any drugs which prolong the QT interval should be reviewed immediately.; and
- QTc is abnormally short if < 350ms.
A useful rule of thumb is that a normal QT is less than half the preceding RR interval.
Causes of a prolonged QTc (>440ms) include:
- Hypokalaemia;
- Hypomagnesaemia;
- Hypocalcaemia;
- Hypothermia;
- Myocardial ischemia;
- ROSC Post-cardiac arrest;
- Raised intracranial pressure;
- Congenital long QT syndrome; and
- Medications / Drugs.
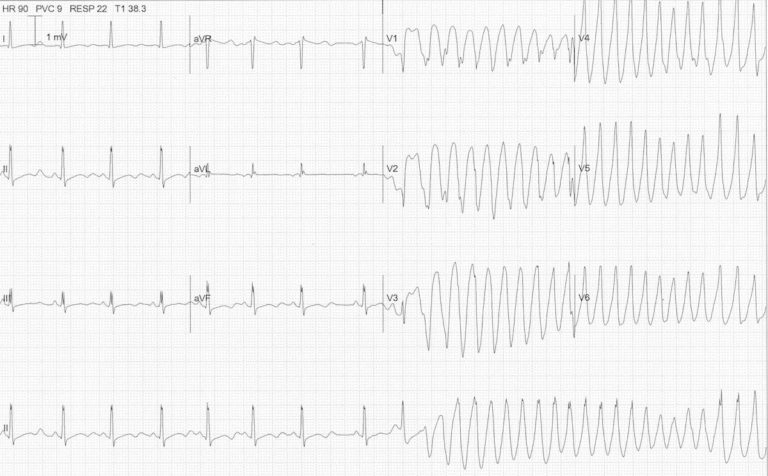
The prolonged QT interval is both widely seen and associated with the potentially lethal rhythm, Torsades de Pointes (pictured). While it can occur spontaneously in the congenital form, there is a wide array of drugs that have been implicated in the prolongation of the QT interval. Paramedics should be aware of the drugs that have been implicated in prolonging the QT interval, such as ondansetron, especially if the patient is already taking a drug which prolongs the QT interval or has a condition associated with QT prolongation.
Most drugs known to cause QT prolongation block the rapid component of the delayed rectifier potassium channel. This prolongs the action potential and lengthens the QT interval. (Delayed ventricular repolarisation will lead to early after-depolarisations, which can result in re-entrant pathways or focal activity and Torsades de Pointes. While many drugs have been implicated in QT prolongation, the actual risk of this occurring is unclear in most cases. The following table lists common drugs which cause QT prolongation and have been associated with torsades de pointes.
https://crediblemeds.org/pdftemp/pdf/CombinedList.pdf
There are three mechanisms by which drugs can interact and increase the risk of QT prolongation:
- Pharmacodynamic Interaction: The concurrent use of more than one drug that prolongs the QT interval increases the risk of torsades de pointes and ventricular arrhythmia;
- Pharmacokinetic Interaction: Some drugs which do not prolong the QT interval themselves can increase the risk of QT prolongation by affecting the metabolism of drugs that do. Commonly used examples of this include drugs such as macrolide antibiotics and antifungals which inhibit the CYP3A4 enzyme. (It has been estimated that CYP3A4 metabolises about half of all drugs on the market and is the same enzyme which grapefruit juice inhibits); and
- Effects on Electrolytes: Hypokalaemia and hypomagnesaemia can increase the risk of QT prolongation, for example, diuretics can interact with QT prolonging drugs by causing hypokalaemia.
QT prolongation is also associated with a number of pathological conditions, including electrolyte abnormalities (hypokalaemia, hypocalcaemia, hypomagnesaemia), cardiac ischaemia, cardiomyopathies, hypothyroidism and hypoglycaemia.
Brief episodes of self-terminating polymorphic ventricular tachycardia do not usually require any specific treatment apart from withdrawal of the suspect drug and correction of metabolic abnormalities. If Torsade de Pointes has haemodynamic consequences it requires prompt termination – cardioversion, or defibrillation in the pulseless patient, is usually effective. Infusion of magnesium or acceleration of the heart rate with rapid pacing or isoprenaline infusion can be useful as stabilisation therapy in the acute setting. To prevent a recurrence the offending drug should be withdrawn and any electrolyte abnormality is corrected. Patients with proven congenital or idiopathic long QTc syndrome who have a history of cardiac arrest, syncope, documented Torsade de Pointes or a family history of sudden death at a young age are often treated with an implantable cardiac defibrillator.
Further reading:
Podcast:
References:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4110870/
http://www.ggcprescribing.org.uk/media/uploads/ps_extra/pse_21.pdf
https://www.mja.com.au/journal/2017/207/3/how-measure-qt-interval
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4040880/
https://www.cidg.org.nz/wp-content/uploads/2017/08/How-to-measure-a-QT-interval.pdf
https://www.nps.org.au/australian-prescriber/articles/drugs-and-the-qtc-interval
https://www.pharmacytimes.com/publications/issue/2008/2008-09/2008-09-8687