Traumatic cardiac arrest reflections
In the early hours of a weekend morning an ambulance is called for a young adult male involved in a single motorcycle collision. While not raining, the roads were still wet from an earlier rainstorm. The ambulance crew took approximately ten minutes to arrive on scene, where they found a police officer performing chest compressions on the motorcyclist.
Initial assessment identified an adult male with an obvious large head wound accompanied with serious haemorrhage, laying supine in the centre of the road against the curb of the roundabout traffic island, approximately 3m away from the bike’s final resting place. The patient’s helmet had been removed prior to the ambulance arrival; he was unresponsive, apnoeic and pulseless. The patient was rapidly intubated with with a 9.0 ET tube (confirmed by visualisation, auscultation and waveform capnography), had bilateral needle chest decompression, a T-POD® pelvic stabilisation device applied and intra-ossesseous access via the right proximal tibia with fluid challenge (followed by adrenaline) commenced as chest compressions were continued and asystole was confirmed on the monitor.
Initial EtCO2 values were in the region of 30-40 mm Hg but as the resuscitation attempt progressed they gradually fell to less than 10 mm Hg after 20 minutes. After 20 minutes the decision to terminate the resuscitation attempt was made.
Reflecting on the incident and management, in conjunction with reviewing the relatively new Australian Resuscitation Council Management of cardiac arrest from trauma guideline, we ask if anything could have been done differently to change the outcome. While most deaths due to trauma occur in the first five minutes after the traumatic event, with most of these unpreventable, recent studies have identified that survival from cardiac arrest is possible – showing between 5.1 to 7.5 per cent survival to discharge – so resuscitation of traumatic arrest is not be considered futile and should always be attempted (unless injuries are incompatible with life).
Management of a traumatic cardiac arrest focuses on the three common causes of preventable early death in trauma, namely haemorrhage, airway obstruction and tension pneumothorax. The exception to this algorithm is the medical patient who had a cardiac arrest before undergoing a traumatic event, for example a patient with chest pain and subsequent VF arrest who then crashes their car or falls from a ladder. These patients are unlikely to benefit from the traumatic arrest management algorithm and would likely do better with traditional Advanced Life Support.
Hypovolaemia
Considering the large quantity of blood on the road surface, in combination with the open head injury and haemorrhage from the ear, it was obvious that this patient had lost a significant quantity of blood. Now in cardiac arrest, there was minimal bleeding but dressing the open wound and application of a pelvic binder should stem or at least slow bleeding on restoration of spontaneous circulation.
IO access was obtained (proximal tibia) and a fluid challenge (normal room temperature saline as indicated by clinical guidelines). In hindsight, choosing the humeral head would have achieved a more rapid fluid administration rate than the proximal tibia and/or a second IO cannulae could have been placed – flow rates 3-4 times quicker have been identified when comparing the humerus to proximal tibia. As IO education had previously focused on the proximal tibia as the primary insertion site, it is understandable why this site was selected by the paramedic.
Hypoxia
According to the guidelines, a patient in cardiac arrest in trauma should have the airway opened as quickly as possible while not delaying blood volume expansion and relief of a possible tension pneumothorax. While a chin lift and/or jaw thrust are the preferred techniques, according to the guidelines, medium-to-long term maintenance of the airway will require a more definitive airway, especially if the patient could be transported to ED.
As the case involved a multiple ambulance response, there were several paramedics on scene, which allowed airway management, venous access and fluid therapy to be achieved simultaneously. Apart from the traumatic injuries and pre-hospital environment, the patient did not appear to have anatomy indicating a possibly difficult airway which, combined with a possible requirement for higher ventilation pressures due to a tension pneumothorax (as indicated by mechanism), the decision was taken to intubation the patient. This was successfully achieved with a size 4 Macintosh blade and a 9.0 ET tube on the first attempt with no interruptions of the chest compressions. For organisations that have adopted ‘finger thoracostomy’ rather than needle decompression (see below), then intubation and mechanical ventilation needs to accompany finger thoracostomy to prevent air entering the pleural space.
There has been an increase in focus on human factors in relation to airway management in the last decade, including the use of checklists and algorithms to reduce risk and improve clinical care. in this case, a physical checklist was not utilised, but the airway management clinical decision making process, equipment required and failed/ difficult intubation procedure was considered prior to the intubation attempt – more of a mental checklist. Below Scott Weingart describes his EMCrit Intubation Checklist.
Tension pneumothorax
Bilateral needle chest decompression of the motorcyclist resulted in a hiss of air from the left needle, but no visual or audible confirmation of a tension pneumothorax for the right side of the chest. This could have been due to there only being a left tension pneumothorax rather than bilateral or that the needle chest decompression was unsuccessful. The cannula used was an 83 mm long 14 gauge cannula, which was sufficiently long enough to reach the pleural space in a patient with this body size.
According to the guidelines, finger thoracostomy is the preferred method of chest decompression, however, needle chest compression can be considered as a rapid alternative in some circumstances. Traditionally, the location for cannula insertion was the second intercostal space, mid-clavicular line, immediately superior to the third rib, however it has been suggested that the optimal location is unknown and will be dependent on individual body habitus.
It is not just the Australian Resusciation Council that recommends alternatives to traditional needle chest decompression. LITFL suggested a while ago that it could be time to change the traditional treatment for a tension pneumothorax as needle chest decompression in the second midclavicular line, intercostal space is only successful in 50 per cent of cases – Tension pneumothorax – time to change the old mantra? The authors suggest that the 5th intercostal space mid axillary line is used for emergency decompression, especially as this is widely accepted as the location for chest drains, there is a thinner chest wall (increased chance of successful access to the pleural space) and anatomically there are no major blood vessels nearby.
A finger thoracostomy involves incising 3-4cm of skin over the 4th intercostal space immediately anterior to the mid-axillary line, followed by blunt dissection into the pleura to allow introduction of a finger into the pleural space. For those unaware of what a finger thoracotomy entails, the following video demonstrates the process.
Chest compressions: a lower priority
In this instance, the presence of a police officer performing CPR allowed the paramedics to control haemorrhage, open the airway along with attempting to restore circulating fluid and relieve tension pneumothorax. Without bystander CPR, these potentially reversible causes should have priority over chest compressions – after years of practice and education emphasising the importance of chest compressions, it can be difficult to change the mindset to defer chest compressions.
Considering patients in cardiac arrest caused by hypovolaemia, cardiac tamponade or tension pneumothorax, chest compressions are unlikely to be as effective as in normovolaemic cardiac arrest. This is the reason that chest compressions take a lower priority than the immediate treatment of reversible causes. In an out-of-hospital setting, only essential life-saving interventions should be performed on scene and rapid transfer to the nearest appropriate hospital should be considered.
The view of chest compressions having no role in traumatic cardiac arrest is not necessarily new, back in 2013 ERCAST suggested just that and was discussed further in 2015 – No CPR in trauma arrest.
Personally, listening to the inspirational Dr John Hinds (RIP) describe the management of a successful traumatic cardiac arrest (starts at 14 minutes into the below presentation, but the entire video is worth a watch) where initial response did not necessarily involve chest compressions was my realisation of the importance of managing the three common causes of preventable early death in trauma – namely airway obstruction, tension pneumothorax and serious haemorrhage, – before commencing chest compressions. Of course, in his circumstances, having a response time of approximately 30 seconds after the traumatic event was also beneficial.
Rapid transport or on scene resuscitation?
The incident occurred 15 minutes (priority one driving) from the nearest emergency department and 20 minutes from a trauma centre. It has been argued that as there are several potentially effective interventions, including resuscitative thoracotomy, available only in hospital for a patient with a traumatic cardiac arrest, then rapid transportation should take precedence over pre-hospital interventions. It should be noted, however, that the highest survival rates after traumatic cardiac arrest are seen in circumstances where the patient was transported after the restoration of spontaneous circulation. Unless the patient is thought to require an emergency department thoracotomy and the patient can be transported there within 10 minutes, according to the Australian Resuscitation Council guidelines, transport should only occur after the return of spontaneous circulation.
While the trauma centre could undertake a resuscitative thoracotomy, the earliest the patient could have arrived there would be approximately 40 minutes after the impact – clearly longer than recommended 10 minutes.
As the cardiac arrest occurred out-of-hospital and ROSC was not obtained, the patient could not be considered for donation after cardiac death (DCD). In Australia and New Zealand, only Maastricht category 3 and 4 patients are regarded as suitable for DCD. The Maastricht criteria were defined in 1995 to categorise potential DCD donors:
Category 1: Dead on arrival at hospital — unknown Warm Ischaemia Time (WIT): ‘Uncontrolled’.
Category 2: Failed resuscitation (in emergency department or ICU) — known WIT: ‘Uncontrolled’.
Category 3: Withdrawal of treatment in ICU — known and limited WIT: ‘Controlled’.
Category 4: Cardiac arrest following formal determination of brain death but before planned organ procurement — known and potentially limited WIT: ‘Uncontrolled’.
Categories 1 and 2 are considered ‘uncontrolled’ and are unlikely to be contemplated for DCD, due to the ethical and logistical difficulties of ‘protecting’ the organs while obtaining consent from grieving family members, and due to the prolonged or unknown WIT, according to the Australian and New Zealand Intensive Care Society (ANZICS)’s statement on death and organ donation. Information is also available regarding the National Protocol for Donation after Cardiac Death and there is also a Professional education package.
Termination of resuscitation
There is no consensus on how long the resuscitation attempt should continue, especially as restoration of a circulating blood volume sufficient to sustain circulation can take several minutes. Once potentially reversible causes have been addressed, then it is suggested that BLS or ALS be continued for up to 10 minutes. Prolonged (greater than 10 minutes) cardiopulmonary resuscitation, after reversible causes have been addressed, is almost never associated with a good outcome. Studies have also suggested that an EtCO2 value of less than 10 mm Hg was accurate in predicting death in patients with a traumatic PEA.
Teamwork
The cardiac arrest was managed by one paramedic directing the other clinicians on scene, with individual clinicians each applying the pelvic binder and haemorrhage control, managing the airway and ventilation, and another controlling fluid therapy and adrenaline. This team leader approach was adopted by the paramedic after witnessing, and participating, in well-managed in-hospital resuscitation attempts. This resuscitation team dynamic approach, is one step towards the pit crew principles, named after Formula One pit crews, which has gained publicity in recent years. The pit crew principle involves team members using a highly choreographed approach to complete individual tasks in parallel with virtual autonomy – during resuscitation attempts, the team leader assigns crew members to discrete therapeutic tasks that are performed without further direction from a team leader. To be effective, all team members have to understand and be confident in every individual role.
Looking towards the UK’s Resuscitation Council guideline for advice on the management of an out-of-hospital cardiac arrest, resuscitation requires a system to be in place to achieve the best possible chance of survival, including both technical and non-technical skills (teamwork, situational awareness, leadership, decision making). To achieve this, there has to be an allocation of roles:
- Appoint a team leader as early as possible; ideally they should be a paramedic or clinician experienced in prehospital resuscitation;
- The team leader should assign team members specific roles, which they clearly understand and are capable of undertaking. This will promote teamwork, reduce confusion and ensure organised and effective management of resuscitation;
- Minimum of four trained staff is required to deliver high quality resuscitation. This will necessitate dispatch of more than one ambulance resource;
- Ensure there is 360° access to the patient (‘Circle of Life’):
– position 1: Airway (at head of patient) – the person must be trained and equipped to provide the full range of airway skills.
– position 2: High quality chest compressions and defibrillation if needed – at patient’s left side. Be prepared to alternate with the operator at position 3 to avoid fatigue.
– position 3: High quality chest compressions and access to the circulation (intravenous, intraosseous) – at patient’s right side.
– position 4: Team leader – stand back and oversee the resuscitation attempt, only becoming involved if required. The team leader should have an awareness of the whole incident and ensure high quality resuscitation is maintained and appropriate decisions made.
Cliff Reid at a SMACC conference conveyed the core concepts of an optimal resuscitation, especially describing the approach of the resuscitation team leader. While directed towards in-hospital teams, the principles can also be applied to out-of-hospital.
Debriefing
After the incident, due to operational requirements, there was no opportunity for a debrief. The back-up crew cleared scene and were given another emergency call. The primary crew returned to the ambulance depot to clean and restock the ambulance in preparation for the next call. In hindsight, in opportunity to explain the reasoning for the clinical decisions made during the resuscitation attempt might have been beneficial, as there was a risk that the self-nominated team-leader was being overly dictatorial.
Following an out-of-hospital resuscitation attempt, an immediate debrief with the attending team can be invaluable, not only providing a learning opportunity, but also ensuring the welfare of the pre-hospital team, especially if the patient is of a young age. According to the UK Resuscitation Council, debriefing in a structured manner is particularly useful to explore non-technical elements of resuscitation such as communication, leadership and team working. Ideally, the debrief is led by a person trained and capable of leading the session in a non-threatening, constructive manner.
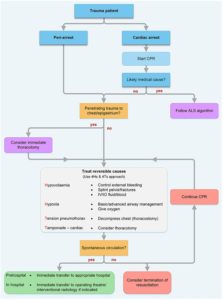
Algorithm
The UK’s Resuscitation Council’s 2015 guidelines introduce a pre-hospital traumatic arrest algorithm. According to the UK Resuscitation Council, adherence to the key steps in this algorithm, which aims to rapidly identify and correct reversible causes of traumatic cardiac arrest, can lead to survival.
The European Resuscitation Council have a similar algorithm, which when compared with the Greater Sydney Area HEMS’s Standard Operating Procedure (SOP) for traumatic cardiac arrest, is consistent with their operating procedures on traumatic cardiac arrest and haemorrhage control. Further information on their SOP and the guidelines can be found in Cliff Reid’s blog article.